This paper by Claassen et al., 2019 looks at EEG pattern changes in response to verbally given movement commands to see if there is a subset of vegetative state patients who are cognitively responsive and yet who have no motor response. The hope is that this might predict eventual outcome.
This paper by Claassen et al., 2019 looks at EEG pattern changes in response to verbally given movement commands to see if there is a subset of vegetative state patients who are cognitively responsive and yet who have no motor response. The hope is that this might predict eventual outcome.
The study took 104 patients who had had acute brain injury. Most (85%) had non traumatic brain injury, which in general carries a more predictably bad prognosis. These patients were either in a vegetative state or in a somewhat better minimally responsive state, e.g. localising to pain but not obeying commands.
The EEG testing was performed within a few days of initial ITU referral.
In a trial, a patient was asked eight times to open and close their hand repeatedly for 10 seconds and then relax their hand for 10 seconds while recording ongoing EEG activity. Two second time blocks were analysed in the frequency domain by calculating the power spectral density (PSD), looking at the relative strength of signal in each EEG lead in four different frequency ranges (delta, theta, alpha and beta).
A “machine learning algorithm” was used to distinguish the “move” PSDs from the “stop moving” PSDs.
Patients were considered to show EEG activation if the algorithm consistently showed a significantly greater than chance (p=0.5) level of ability to distinguish moving command to stop moving command.
Outcome was determined by the standard Glasgow Outcome Scale after 12 months, with values >=4 (being able to be left up to 8 hours alone) defined as a good outcome.
Ultimately, patients who had at least one record showing EEG activation had a 44% chance of good outcome as defined above and only 14% of patients without EEG activation had a good outcome (with 5% missing data).
Discussion
Some of the patients were under some sedation for safety reasons, which could influence their responsive in a more reversible manner unrelated to their brain injury and also affect their EEG, although this would be unlikely to affect the change in pattern of EEG over several seconds, other than through the patient’s genuine response level.
It might have been worthwhile to record surface EMG of the forearm flexors, just to confirm there was no difference in EMG activity between “EEG activation” patients and those with no EEG change. In a patient with critical illness neuromyopathy, a little movement or muscle activation might not easily be seen.
Because patients were just taken consecutively, rather than being matched according to their coma severity, there could be poor matching and this was indeed present, where the patients who were subsequently found to be “EEG responsive”, and eventually to have a better outcome, were less likely to be in the worst comatose category at initial enrollment (50% vs 55%) and more likely to be in the best category (31% versus 23%). Although the odds ratios were not statistically significant, this does not mean that with any degree of confidence there was positive evidence for no difference in initial severity between the groups.
In fact, if one stratified patients according to the initial three clinical severity categories, would that have more powerfully predicted better outcome than “EEG responsive” or not, making the test redundant?
On technical appraisal of the methodology, it seems that the power spectral densities were individual 2-second blocks, with all the comparisons and averaging being done subsequently by the machine learning pattern recognition algorithm.
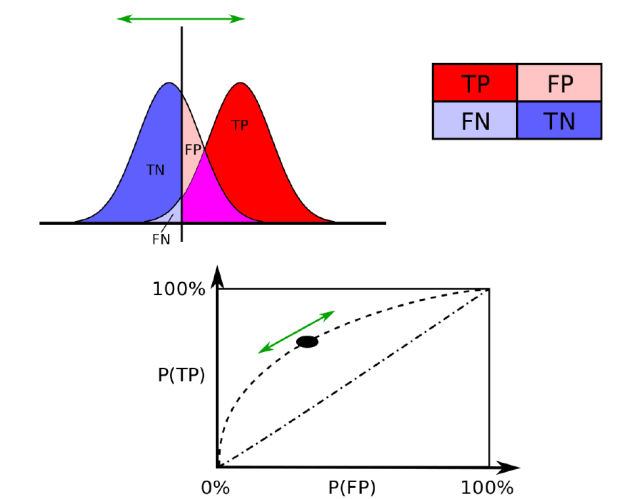
Statistically, the paper used the single value of the area under the curve (AOC) of the receiver operating characteristic (see below). This means that across a range of sensitivities (or true positives (TP), where the algorithm correctly decides that there is enough of a difference between the “move” and “stop moving” patterns), there is an opposing range of false positives (FP). How convex is the curve that describes this range relates to how good the test is. A value of 1 means perfect classification, 0.5 is just random (the straight diagonal in the figure below), and 0 means the pattern change is actually reliably identifying the stop pattern when it was supposed to identify the move pattern.
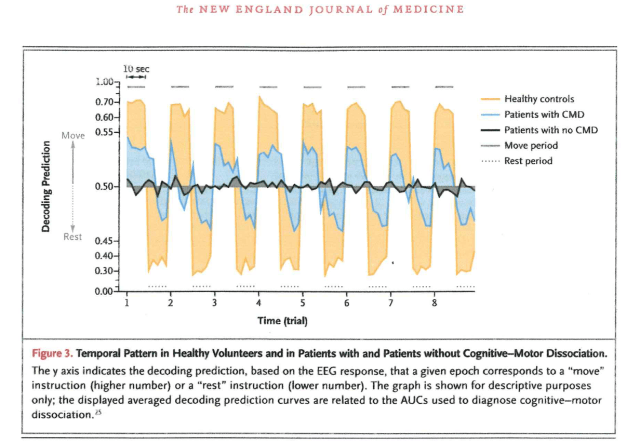
This is shown in their fig. 3 (below), which seems to show the AOC values for each of the 5 “move” 2-second samples (hence the varying level across each peak and trough) followed by each of the 5 “stop moving” samples, with the whole thing repeated 8 times. However, they say that the graph is shown “for descriptive purposes only” so we do not know how it relates to the real data! We do not know if these are actual averages for all the controls, all the EEG responsive patients (which they call cognitive motor dissociation (CMD)) and all the non EEG responsive patients. If they are averages, they would have to be across all the first 2-second epochs and then all the second 2-second epochs, etc.
Where this is important is that although the algorithm provides a discrete yes-no answer, the confidence of this answer is a continuous variable, and there is a suspicion that this confidence level may fall into a continuous range with healthy volunteers at one end and the most unresponsive EEG patient at the other, rather than there being three discrete modal peaks of normal, EEG responsive and EEG unresponsive. If the former, the inevitable variability about a single mode makes the test far less useful as a predictor of outcome in individual patients. At best, it could be an independent predictor that, combined with other predictors, could build up a reasonably confident prognosis.
A major issue with patients in a vegetative state is when to withdraw support. In the UK, in patients with non traumatic acute brain injury, persistent vegetative state is defined as such around 3 months after injury and this is the time when conversations may be had along these lines on the basis that if the patient has not “woken” by this time, the chance they may eventually do so, with a reasonable quality of life, becomes remotely slim. No-one is ever going to think about withdrawing support at 6 days post-injury on the basis of an “EEG unresponsive” result.
This Journal Club post was presented by Dr Rubika Balendra, Specialist Registrar in Neurology at Barking Havering and Redbridge University Hospitals NHS Trust.