Background
Background
Enteroviruses, which may cause gastroenteritis or upper respiratory tract infections, are well-known to have neurotropism – a predilection in a proportion of individuals to spread to certain types of neurones, thereby resulting in characteristic neurological syndromes that occur after initial infection. The most well-known example is of course poliomyelitis which results in acute flaccid paralysis from anterior horn cell involvement. More recently, enterovirus A71, causing hand foot and mouth disease, has resulted in epidemiological clusters of brainstem encephalitis and acute flaccid paralysis.
Enteroviruses may also be responsible for acute viral meningoencephalitis, presenting classically with fever, meningism and obtundation, and perhaps with focal cerebral neurological signs or seizures. In fact, in our experience, enteroviruses, along with herpes simplex virus, are the most commonly identified organisms responsible for sporadic viral meningoencephalitis.
This paper, by Messacar et al. in the Lancet (2015), reports cases of the latest enterovirus identified to have possible neurotropism, namely enterovirus D68. Localised outbreaks occurred between 2008 and 2010, and a large outbreak occurred in the USA in the Autumn of 2014. A cluster of cases of acute flaccid paralysis in children were identified at the same time (Autumn 2014); they presented with weakness in proximal limb, facial and bulbar muscles and radiological changes of either longitudinal grey matter spinal cord lesions and/or brainstem hyperintensities were identified.
Study Design and Findings
Cases were retrospectively identified from records of children admitted to a hospital in Colorado in the Autumn of 2014. Inclusion criteria were acute flaccid paralysis with mainly grey matter spinal cord involvement on imaging and/or acute cranial nerve dysfunction with brainstem lesions on imaging.
In the acquisition period twelve children satisfied the neurological inclusion criteria. All but one had fever and preceding upper respiratory tract symptoms. Ten had meningism. The limb weakness, present in ten cases, was proximal with hyporeflexia and preserved sensation. A similar proportion of children had symmetrical and asymmetrical weakness. Ten had cranial nerve dysfunction, bulbar weakness, diplopia or facial weakness. None had encephalopathy or seizures.
The spinal cord lesions, present in eleven children, always affected the central grey matter, especially the anterior horn cells. The longitudinal spread was from 4 to 20 vertebral levels (we commented that there were only 19 levels from C1 to T12, but younger children’s spinal cords extend a little lower). Brainstem lesions were present in nine children, mainly in the dorsal pontine tegmentum.
Some children had EMG, which showed variable motor denervation, presumably indicating either anterior horn cell or ventral nerve root involvement.
Spinal fluid analysis typically showed a pleocytosis (unlike typical cases of acute motor axonal neuropathy subtype of Guillain Barre syndrome) and normal or mildly elevated protein in all but one case.
In five of eleven cases, nasopharynx specimens were positive for enterovirus D68. Blood and spinal fluid PCR was negative, as is commonly the case in polio and other enterovirus neurotropic infections. Polio also has a delay between initial and neurological symptoms and yet is due to direct viral spread to the anterior horn cells, so the delay after respiratory symptoms and the lack of spinal fluid virus observed in these cases does not necessarily indicate that the condition is post-infectious rather than infectious.
Many of the children were treated with intravenous immunoglobulins or steroids. Some had plasmapheresis. There was no clear benefit from these therapies. Many children had lengthy admissions and significant residual neurological deficits despite treatment.
The paper compared the number of these neurological cases in 2014 with average numbers of similar presentations (from ICD 9 code discharge database) between 2010 and 2014. In any previous 3 month period the maximum number of cases was 4 (significantly lower). One was positive for D68.
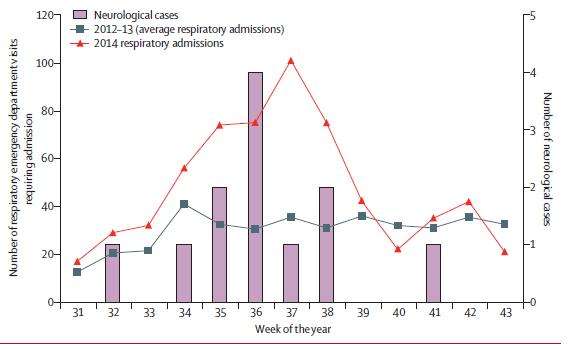
The background epidemiological context in the paper describes a 77% increase in children with acute respiratory admissions in 2014 compared with equivalent months in previous years. It is not clear if all such cases were tested for viruses but a number were tested with a screening nasopharyngeal swab PCR array that does not distinguish enteroviruses from rhinoviruses. There was a “substantial increase” in positive results on this test during the acquisition period. Only 25 cases were actually tested for D68, and these were cases admitted to ITU with severe respiratory disease. These were positive for D68 in 76%.
Opinion
The paper presents a persuasive argument for a defined neurological syndrome, with the flaccid paralysis, exclusive motor involvement, pleocytosis and rather characteristic neuroimaging features.
Less persuasive is the causative role of enterovirus D68. It was identified in less than half of the neurological cohort during a presumptive D68 outbreak. The background level of enterovirus D68 positive asymptomatic children during the acquisition period is unknown. The increase in neurological cases during that Autumn epidemic compared to baseline levels could be skewed by ascertainment bias and, given the sample size, a coincidental increase is always possible.
Neurological D68 infection is not new. In fact a previous paper (Ayscue et al., 2014) (which this paper mentions at the end of the discussion) reported 23 cases of acute flaccid myelitis in California from 2012 to 2014, two of which were positive for D68. The lower rate of D68 identification could have reflected testing late during the illness, whereas earlier testing as in this paper is more likely to yield positive results.
Nevertheless the paper does contribute to a body of evidence suggesting that enterovirus D68 is one of a group of viruses that have accounted for recent outbreaks of upper respiratory tract infections and may have particular patterns of neurotropism resulting in acute infectious or post-infectious complications. Unfortunately, as the paper identifies, there are as yet no specific and effective preventative or treatment strategies for these neurotropisms.
This paper was presented to our Journal Club by Dr Sian Alexander, Specialist Registrar in Neurology, Queens Hospital, Romford, UK.