 Stroke, defined as a sudden vascular event resulting in localised brain damage (World Health Organisation, 1978), is without doubt a major challenge in health care, being the third most common cause of mortality in developed countries and the single greatest cause of lasting disability (Mant et al., 2004). In the UK, stroke patients occupy 2.6 million days in hospital beds a year, equivalent to one in five total acute hospital beds and one in four long-term beds (National Audit Office, 2005). Over the last decade, there have been increasing efforts to organise acute stroke care into dedicated stroke units and to raise public awareness that stroke is a medical emergency to be managed in a timely fashion (e.g. the FAST campaign).
Stroke, defined as a sudden vascular event resulting in localised brain damage (World Health Organisation, 1978), is without doubt a major challenge in health care, being the third most common cause of mortality in developed countries and the single greatest cause of lasting disability (Mant et al., 2004). In the UK, stroke patients occupy 2.6 million days in hospital beds a year, equivalent to one in five total acute hospital beds and one in four long-term beds (National Audit Office, 2005). Over the last decade, there have been increasing efforts to organise acute stroke care into dedicated stroke units and to raise public awareness that stroke is a medical emergency to be managed in a timely fashion (e.g. the FAST campaign).
The development of thrombolysis has been one of the drivers for management of stroke as an emergency. This “clot buster” treatment may be given intravenously to dissolve the thrombus or embolus in a cerebral artery and allow reperfusion of the territory supplied by the artery before those areas of the brain become irreversibly infarcted. Timing is critical for such treatment to be effective; if given too soon after symptom onset, the thrombolysing agent (tissue plasminogen activator (TPA)) may be unnecessary as the patient may in fact be suffering a transient event that would reverse spontaneously, and if given too late the brain tissue will already be dead.
 The standard European criteria for thrombolysis, developed from the major multicentre study (Safe Implementation of Thrombolysis in Stroke Monitoring Study, abbreviated to SITS-MOST (Lancet. 2007)) that validated its use, originally stipulated a time window of 3 hours after symptom onset and excluded patients whose symptoms were rapidly resolving. In practice, giving the treatment too early is not a major concern as most self-resolving events, called transient ischaemic attacks (TIAs) last less than an hour and it is very rare to be ready logistically to thrombolyse within an hour of symptom onset.
The standard European criteria for thrombolysis, developed from the major multicentre study (Safe Implementation of Thrombolysis in Stroke Monitoring Study, abbreviated to SITS-MOST (Lancet. 2007)) that validated its use, originally stipulated a time window of 3 hours after symptom onset and excluded patients whose symptoms were rapidly resolving. In practice, giving the treatment too early is not a major concern as most self-resolving events, called transient ischaemic attacks (TIAs) last less than an hour and it is very rare to be ready logistically to thrombolyse within an hour of symptom onset.
Outcome following Thrombolysis
Unfortunately, thrombolysis is not a panacea even within this narrow time window. A fair comparison is achieved with a randomised double-blind study against placebo, but because of widespread use such studies have not recently been performed. The original positive trial (National Institute of Neurological Disorders (NINDS) Stroke Study Group, 1995) showed no clear clinical differences after 24 hours but what was described as “at least 30% better outcome” at 3 months (global odds ratio 1.7 (95% confidence interval 1.2 to 2.6). By way of example, the percentage of patients achieving 0-1 on the modified Rankin score (meaning no or minimal disability) was 39% vs 26%, in other words 13% more patients had excellent outcome after thrombolysis than after placebo. There was no improvement in mortality.
However, there have been concerns over the fact that in this study the placebo patients had a worse severity stroke at onset, that other studies have shown unclear benefit, and that some studies have relied upon open label self-reporting by patients to measure outcome.
Underpinning these concerns is the risk of haemorrhage associated with intravenous thrombolysis. Thrombolysis was originally developed for coronary thrombosis in myocardial infarction; the brain is an organ far more sensitive to insult and reperfusing infarcted brain may make it particularly susceptible to haemorrhage, with far worse consequences than a haemorrhage into myocardium. In the NINDS study, 7% of patients had a symptomatic intracerebral haemorrhage (meaning neurological deterioration or other clinical suspicion in presence of haemorrhage on CT not seen pre-treatment) within 36 hours after thrombolysis, versus 1% of patients given placebo. In 3% of thrombolysed patients, the haemorrhage was fatal.
The SITS-MOST study was designed to look at safety of thrombolysis given according to the same protocol and collected data on 6483 patients and found a similar figure of 7.3% patients significantly worsened (<=4 points higher on NIHSS score) within the first 7 days by intracranial haemorrhage.
So when counselling patients on giving thrombolysis, we should say that within the 3 hour window, out of 100 treated patients, around 12 will have a better outcome (more likely to be disability free or minimal disability), 4 will be made worse because of brain haemorrhage, 2 will die from brain haemorrhage and 82 will be unchanged. It does not sound as good as quoting 30% better outcome (taken as the increased proportional percentage gain rather than absolute percentage gain over placebo).
Recent Changes to Prescribing Guidelines
More recent studies have explored widening the window of thrombolysis to 4.5 hours, or even longer in certain circumstances. The third European Cooperative Acute Stroke Study (ECASS III, 2008) randomised patients at 3 to 4.5 hours after stroke onset to thrombolysis or placebo, and found a 52.4% versus 45.2% good outcome; the significance level for this 7% improvement was only 0.04 – the lower 95% CI for the odds ratio of better outcome (according to their chosen criterion on Rankin) was 1.02! If other Rankin criteria were chosen, e.g. 0-2 versus 3-6 instead of 0-1 versus 2-6), no significant improvement would be demonstrated. In fact the chance of being dead or severely disabled at 3 months (modified Rankin scores 5-6) was non-significantly higher if thrombolysed (14.8% versus 13.4%). Concern has also been voiced that, despite randomisation, the placebo group had on average a more severe stroke before thrombolysis (one point worse on NIHSS), and were more likely to have had a previous stroke. The risks of intracerebral haemorrhage were comparable to data from patients thrombolysed within 3 hours.
In counselling a patient within this time window, we would therefore have to add that because the time is more than 3 hours after thrombolysis, the chance of improvement to the state of no or minimal disability increases by around 7% instead of 12%.
Cost effectiveness analysis of patients thrombolysed in this time window show limited favourability but are based on limited evidence; of course fatalities reduce cost compared to disability so I find such analysis morally inappropriate.
In the UK, the National Institute of Clinical Excellence (NICE) guidelines for stroke were updated in 2013 to increase the thrombolysis window from 3 hours to 4.5 hours. There is also now no exclusion of posterior territory infarction and debate over excluding patients over 80 years. I personally have reservations about this, and consider it a situation where we have permission under licence to give it if we feel in our judgement it is clinically appropriate. Despite the trumpeting of trial data, there are ethical reservations about giving a treatment that will help a modest proportion of patients but harm a significant proportion too. The haemorrhage risk is more than those surgeons and anaesthetists typically quote for surgery.
Unsurprisingly, this situation polarises medical opinion. Outcome data on thrombolysis have come under intense scrutiny and been subjected to endless meta-analysis and debate. What is really needed is less spin on statistics and more information on predicting a good outcome of thrombolysis in an individual patient who has just had a stroke.
Current guidelines for selecting patients for thrombolysis depend on a clinical diagnosis of ischaemic stroke, a clinical scale of stroke severity, various exclusion criteria and a CT scan of the head. This CT scan will not demonstrate the stroke; the changes of stroke after 3 hours are too early to be detected on CT, which is simply showing the reduced density of infarcted brain. Instead, the CT excludes a haemorrhagic stroke where thrombolysis would be pointless and dangerous, or an established large stroke where thrombolysis may be too late and also associated with increased risk.
An alternative investigation that positively diagnosed stroke within the thrombolysis time window would be very useful to exclude “stroke mimics”, such as patients with acute unilateral muscular weakness from spondylosis or patients who are imagining that they are having a stroke and reproducing its clinical features (functional stroke). What would be even more useful is an investigation that could positively identify ischaemic but potentially retrievable brain from brain that was already infarcted and might only result in haemorrhage if suddenly reperfused by thrombolysis. This retrievable brain is known as the “penumbra”, alluding to the surrounding region of partial rather than complete shadow cast by an object in front of a non-point light source .
CT Perfusion Imaging
Of such investigations, the most promising may be CT perfusion. This requires only the hardware for standard CT, with an intravenous iodinated contrast injection, and may be performed more rapidly and be more easily tolerated than MRI. The limiting factor is likely to be user dependence and the quality of the analysis software.
Technique of CT Perfusion Imaging
After a bolus injection of contrast, a sequence of images are taken that measure the rise and subsequent fall in contrast density as the bolus travels through the cerebral vasculature. Two reference time plots are normally taken; that for the input transit time is the A2 segment of the anterior cerebral artery as it passes perpendicular to the axial plane of imaging, and that for the output transit time is the superior sagittal sinus. For each region of interest voxel, four parameters are then calculated:
- cerebral blood flow (CBF)
- cerebral blood volume (CBV)
- mean transit time (MTT)
- time to peak contrast enhancement (TTP).
These are then mapped onto an axial slice of the brain to convey visually how the different parameters vary across brain regions, with high values represented as red and low values represented as blue.
Flow dynamics tells us that three of these parameters are interdependent:
- MTT = CBV / CBF
So if there is a thrombus reducing flow to a region of brain, as the CBF is lowered, the MTT increases in parallel if the CBV stays the same.
Normal grey matter has a higher CBF and CBV, so cortical areas of gyri tend to look more red on both CBF and CBV. Because the increases are similar, the changes cancel out on MTT so the MTT tends to look more blended between white and grey matter. The venous sinuses also look very red on CBF and CBV and similar to background on MTT, presumably because these voxels are purely blood so have relatively high blood volume as well as flow. (One might expect arteries to have higher flow, and therefore blue on MTT, but the resolution of CT perfusion may not be great enough to identify arteries in cross-section.)
In the early hours after an acute stroke, it is considered that an “umbra” of infarcted brain may be surrounded by a “penumbra” of ischaemic brain that will shortly become infarcted but is potentially salvageable on reperfusion. CT perfusion may allow differentiation of the two because the CBV reduces more in infarcted brain. So:
| Infarcted Brain | ↓↓↓ CBF | ↓↓ CBV | ↑↑ MTT |
| Ischaemic Brain | ↓↓ CBF | slight ↓ CBV | ↑↑ MTT |
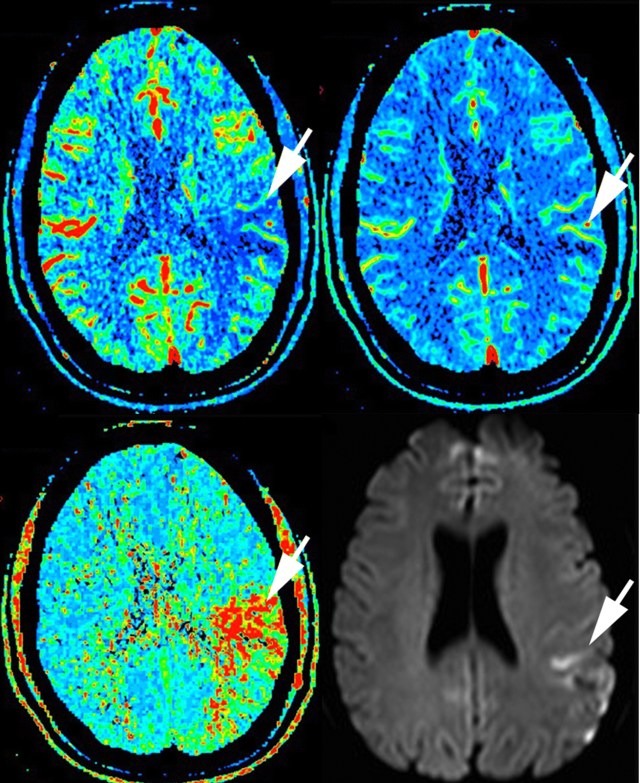
In a patient 70 minutes after stroke onset (NIHSS score 10), the unenhanced CT, not shown, is normal. The cerebral blood flow (top left) and cerebral blood volume (top right) show reductions in the arrowed area. There is a corresponding increase in mean transit time (bottom left) and a DWI weighted high signal area several days later on MRI (bottom right). (Figures taken from Hopyan et al., 2010.)
Cerebral blood flow is obviously reduced if there is a proximal thrombus, and in infarction there is reduction in cerebral blood volume. This could be because of tissue swelling raising local intracranial pressure and constricting capacitance vessels, because there is a certain elasticity in vessels so that constriction will follow from reduced flow, or because of reflex vasoconstriction of capacitance vessels in damaged brain. The reduced CBV is still less than the dramatically reduced flow, so that MTT is significantly prolonged. The infarcted area appears more blue on CBF and CBV and more red on MTT.
In ischaemic brain, the CBV is relatively preserved, perhaps because the affected brain area is not as swollen or perhaps because of preserved reflex capacitance vessel dilatation in an attempt to improve perfusion of these areas. Cerebral blood flow is reduced (blue), but there is now a mismatch between CBV (relatively normal) and MTT (clearly red).
Problems in interpreting CT perfusion
- Image processing is complex and user dependent. There may be poor selection of the anterior cerebral artery and superior sagittal sinus reference points
- If the protocol is poorly designed, the radiation dose may be massive
- Many protocols do not analyse the whole brain, so clinical knowledge is required to determine if the area of interest is middle cerebral artery territory. Brainstem areas cannot easily be assessed.
- The resolution of CT perfusion is such that small strokes may not be visualised
- If there is extracranial vessel occlusion, e.g. carotid artery, the hypoperfused area may give a false impression of acute infarction. The same applies to areas of leukoaraiosis. Thus CT perfusion must be interpreted in the context of unenhanced CT appearances and preferably with CT angiography.
Practical Uses of CT Perfusion
- Identification of penumbra. If a patient was outside the 3-hour time window for thrombolysis, or the time of onset was unknown, but was otherwise a good candidate, a CT perfusion scan revealing a relatively large penumbra with normal CBV and prolonged MTT would indicate salvageable brain that might benefit from thrombolysis.
- Positive identification of stroke. CT perfusion reveals changes very early after stroke onset, but there may be poor sensitivity because of lack of clarity over the territory of interest, the possibility of posterior circulation stroke and poor resolution of a small stroke, e.g. lacunar infarction.
- Measuring cerebrovascular reserve. In the non acute setting, CT perfusion before and after administration of intravenous acetazolamide can help to identify brain areas that are chronically ischaemic. Acetazolamide is a vasodilator, but will have less effect on ischaemic areas because such areas already have ongoing maximal compensatory vasodilation. Thus, after acetazolamide, there will be less increase in CBF in ischaemic areas compared to normal neighbouring areas, less increase in CBV (though this is generally increased throughout the brain), and most clearly an extra prolongation of MTT (more red) in areas that may already have somewhat prolonged times compared to normal areas.
- Identifying vasospasm. In the situation of subarachnoid haemorrhage, areas of brain suffering reactive vasospasm react like the penumbra of a stroke and indicate that measures taken to reduce vasospasm may reduce the risk of lasting focal neurological deficit after subarachnoid haemorrhage.
An accompanying Journal Club Review looks at a study that investigates the use of CT perfusion in acute stroke primarily in terms of stroke diagnosis.

Pingback: Journal Club Review “Certainty of Stroke Diagnosis: Incremental Benefit with CT Perfusion over Non-Contrast CT and CT Angiography | Neurology Online Journal Club