 Introduction
Introduction
For this edition of the Neurology Online Journal Club I wanted to review not one but a series of papers to address a specific issue, namely predicting the risk of seizure recurrence after a single seizure and predicting how much this is reduced by starting anti-epileptic medication. I started with the Multi-centre study of early Epilepsy and Single Seizures (MESS) study, but there is more than one report on the same data set, and its main points prompted a more detailed look at other literature on the subject and my personal views. Hence I have described this as a commentary.
There is an accompanying Journal Club review that deals specifically with risk of seizure recurrence in relation to driving.
Background
Epilepsy is certainly one of the more common conditions managed in neurology and indeed in general medical practice. The lifetime prevalence of seizures (% of people who will have a non-febrile seizure at some point in their lives) is 2-5%, and the prevalence of active epilepsy is around 0.5%. A first seizure often presents as a sudden, shocking event in a previously well person, and often leaves the patient in a similarly well state with the expectation of returning to a reasonably normal life – and yet bewildered and worried. As a result, it is a condition where in my view counselling of the patient regarding management options and involving the patient in decision-making is particularly important.
A specific issue with epilepsy management is that typically there are no ongoing symptoms or abnormal clinical signs. The patient may be starting treatment, exposing them to potential side effects, without making them feel better at all. We may have no idea whether or not the drug is working until it manifestly fails much later in the form of a recurrent seizure, and even then we are not sure what would have happened if we had not started treatment, or had started a different treatment. In this respect epilepsy management is more akin to management of episodic headache or TIA than of Parkinson’s disease or chronic pain.
When management revolves around predicting and minimising risk, statistics inevitably play a part. Clinicians need to have the communication skills to explain clearly to patients in broad terms the likely risks of seizure recurrence in different circumstances, and of course that means knowing those risks and understanding basic statistics themselves. Knowledge of risks is covered in this review, but communicating them remains a challenge. (For example, in the UK a survey revealed that the majority of adults did not appreciate that it was equally likely for one to roll a 6 on a die as any other number, or that a previous coin toss does not affect the result of a subsequent one.)
The key questions to which patients and clinicians need answers are:
- What is a specific patient’s risk of a further seizure over a certain time period? This estimate should factor in whether or not this was their first seizure, the seizure type and aetiology, the time they have already gone without a seizure and other factors that determine risk such as EEG, imaging abnormalities and family history of epilepsy.
- How much is this risk reduced if the patient goes on antiepileptic medication?
- If starting medication, and there are no further seizures, when should this medication be stopped again?
Risk after a first seizure
The FIRST study (First Seizure Trial Group Study) in 1993 reported recurrence risks of 18%, 28%, 41%, and 51% at 3, 6, 12, and 24 months if not given medication, and 7%, 8%, 17%, and 25% if given medication. Randomisation on or off medication was done within 7 days of their seizure, so this is nicely applicable to an “early clinic” or inpatient decision. The odds ratio of reduction of risk by medication was 0.4 (i.e. seizures were only 40% as likely on medication as off medication).
The largest single study on risk of seizure recurrence with randomisation for initial treatment was that conducted by the Multi-centre study of early epilepsy and Single Seizures (MESS) study group; here the risk of recurrence in the 404 randomized to immediate treatment was somewhat lower at 18%, 32%, 42%, and 46% at 6 months, 2, 5, and 8 years after randomization versus 26%, 39%, 51%, and 52% in the deferred treatment group.
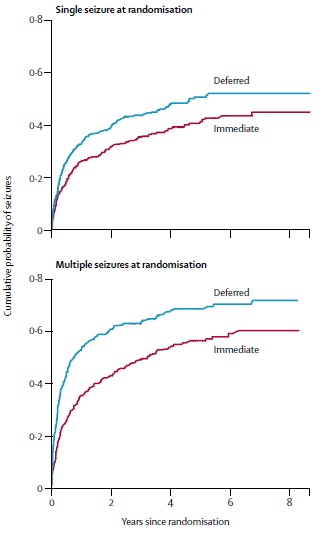
Cumulative risk of recurrence years after a seizure. Note that it is the top figure that specifically refers to a first seizure.
A key difference between the studies is that in the MESS study patients were randomised generally 3 months after their initial seizure. The six month figure is therefore the risk from 3 to 9 months after a seizure, having already gone about 3 months without a seizure.
Further analysis published in the BMJ (2010) of a subgroup of MESS study patients looked specifically at implications for driving and is the subject of a complementary journal club review. This subgroup naturally consisted of those over 16 years of age and those who could have their seizure-free period dated back to their first seizure rather than to time of randomisation; it was found that the 12-month risk of a seizure, having already gone 6 months without a second seizure, was 18% off medication and 14% on medication and this difference did not reach statistical significance.
The lower risk found in the MESS study than in the FIRST study is supported by a prospective study without treatment randomisation (Hauser et al., 1998) and largely on adults; the risk of a first recurrence was 21%, 27%, and 33% at 1, 2, and 5 years after the initial seizure. In those who recurred, the risk of a second recurrence was 57%, 61%, and 73% at 1, 2, and 5 years after the first recurrence. The risk of a second recurrence approached 90% after remote symptomatic seizures (those that are secondary to a brain insult at a previous time and therefore indicating an ongoing risk) and was 60% following cryptogenic/idiopathic seizures.
A problem with comparison and interpretation of study data is in patient selection. While there were 1443 patients randomised in the MESS study, another 404 did not consent to randomisation. Those where the risk might be considered lowest might not want to consider taking medication, while those at high risk might not want to take the chance not to have medication. Furthermore, an actual selection criterion was that for ethical reasons both patient and clinician had to be unsure about whether or not to start medication to be invited to participate.
It is likely that low risk groups in such a study will have overestimated risk, while high risk groups might have underestimated risk and underestimated treatment effect. This possible shortcoming is important in guiding actual practice. If there is a policy from opinion leaders that treatment is not warranted for first seizures, this might get interpreted rigidly by others as a blanket rule and those patients at high risk after a first seizure – the very patients who might not have enrolled on the study – might not even get counselling about the possibility of taking medication.
Finally, different studies may have differing proportions of different seizure type. The MESS study took anyone over the age of 1 year, and there may have been a relatively high proportion presenting with a single minor complex partial seizure.
Decision to treat
Most epileptologists do not treat a single seizure. In fact they define epilepsy as two or more seizures, to try to exclude the significant proportion of individuals who have a single seizure and no further attacks.
Perhaps this conservative strategy is because of the side effects of antiepileptic drugs. These include potential teratogenicity if falling pregnant while on the drug, long-term effects contributing to osteoporosis, possible long-term effects on fertility and possible long-term effects on cognition (mainly mooted in children).
However, there are now many antiepileptic drugs from which to choose, increasing the chance of finding one to suit, and modern drugs may minimise many of these risks. If one looks at the side effects of most drugs taken for any length of time, the list looks at least as scary as that for modern antiepileptics. For example, most anti-migraine drugs also have potential teratogenicity.
If a cardiologist said to a patient who had just had a heart attack, “Well you could have secondary prevention to reduce your risk of a subsequent myocardial infarction (MI) over the next year from 41% to 17% (using the FIRST trial data), but we won’t bother because we don’t really say you have heart disease until you get your second MI”, they would be dialling up for a second opinion before he or she had finished the sentence! And secondary preventatives such as beta-blockers, antiplatelets and statins, and certainly coronary stenting procedures and coronary artery bypass grafts, are not without their risks either.
While the mortality associated with a generalised tonic clonic seizure is lower than that for an MI, it is not insignificant. Quite apart from the circumstances of the attack potentially posing a risk, there is a small but well-documented risk of sudden unexpected death in epilepsy, thought to relate to a number of factors including the extreme autonomic disturbance that occurs during the attack. The event may occur in a young completely healthy person out of the blue, reflects a total loss of self-control, may be potentially embarrassing and stigmatising, and may leave the patient exhausted or potentially even in a psychotic state for days afterwards. I think any trivialisation of a seizure in comparison with an MI can only reflect an age-old prejudice against neurological disease that it is “difficult”, “untreatable” and not suffered by “normal” people.
But other data presented here show that if for some reason an adult patient only saw someone in a position to advise on antiepiletic treatment about six months after their first seizure (the BMJ trial went back to the seizure date not the recruitment date), and they had not had a second seizure in that time, the 12-month seizure risk figures are only 18% vs 14%. This presents a completely different picture of risk of treatment side effects versus reduction of risk of seizures.
Stratification of Risk
Another follow-up to the MESS study (2006) stratified risk of seizure recurrence according to a scoring system (below).
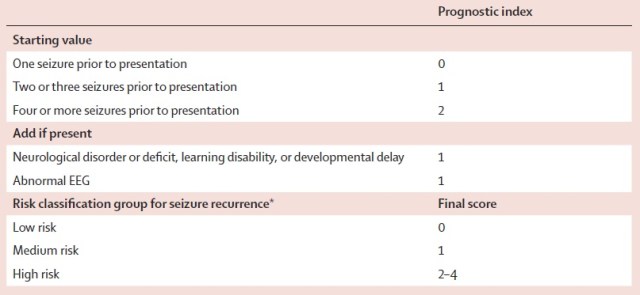
Scoring system for stratification of risk of recurrence after a single seizure according to the MESS study data.
Half of the patients in the MESS study were used to investigate these risk factors to develop the scoring system, and the other half were used to see if subgroups divided post-hoc according to this risk stratification would have differing benefits from medication. It was found that all but the lowest risk subgroup would benefit from medication (see below); in fact it bizarrely seems in the lowest risk category that avoiding treatment is non-significantly protective (p=0.2).
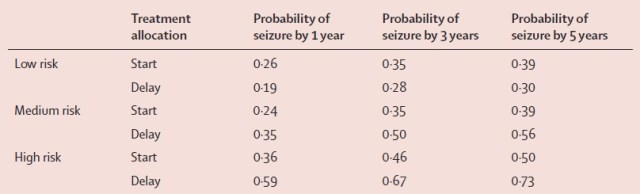
Kaplan-Meier derived estimates of probabilities of seizure recurrence divided according to different risk groups. Start and delayed treatment refers to treatment started at randomisation or delayed until subsequent seizures.
This information could therefore provide a basis for individualising risk assessment and individualising decisions to treat on that basis, or at least providing a default strategy. However, it would be applicable only to patients seen in a clinic fully three months after their seizure who had not already started medication or had another seizure in the meantime.
When to stop treatment
If one is to embark on treatment, perhaps controversially so after a first seizure, when does one stop?
Antiepileptic drugs are probably only protective while being taken. This is indirectly illustrated by long-term remission figures in the MESS trial. Initial treatment decisions did not affect the overall figure of 92% of patients being at least 2 years seizure free 5 years after enrolment. In other words if treatment was deferred until a second seizure, they were as likely eventually to go into remission, but had obviously had more than one seizure while getting to that point and might still be on medication at that point.
One rationale would be to treat for as long as the drug appears from population studies to be significantly reducing the risk of a subsequent seizure.
The longer the patient is seizure-free, the closer data taken from patients with single seizures recruited 3 months late will correspond to those taken immediately, so the more accurate the original MESS data become. We see that from this study’s long term follow-up, the 2 year risks were 32% vs 39%, 5 year risks were 42% vs 51%, and the 8 year risks 46% vs 52%. There is probably a diminishing return over time, but it is difficult to draw a firm conclusion as to significance of this reduced risk at different times.
Most studies specifically looking at timing of antiepileptic withdrawal are on patients who had had more than one seizure, precisely because most clinicians do not start treatment for a single seizure in the first place! Obviously the findings cannot be applied to those who had a single unprovoked seizure, because the overall risk is lower in this group.
One study (JNNP 2002) on patients who mainly had multiple seizures but which at least selected patients on monotherapy, and so tended to reflect patients more easily controlled, found that after 2 years the 12 month recurrence risk was 9% continuing on medication vs 26% stopping medication; on a multivariate analysis, the hazard ratio was 2.6 (CI 1.5 -4.8), and the hazard ratio dropped to 1.6 (1.0 – 2.6) if 3 to five years seizure free and to 1.0 if >5 years seizure free. So after multiple seizures there is clearly no excess risk from stopping medication only if seizure free for >5 years.
Conclusions
Umm…
We have conflicting risks, conflicting risk reductions from medication and data that apply only in specific circumstances.
What we need is a large multi-centre study that:
- Randomises patients immediately, so we can make an informed treatment decision at an appropriate time when the recurrence risk is highest
- Subdivides into age groups, as the paediatric population and geriatric population may have different seizure aetiologies from young adults, and even different clinicians.
- Subdivides according to generalised tonic clonic versus complex partial seizures. The latter are by no means as severe and dangerous, and one might imagine that if the first seizure is complex partial, there may be a higher chance of a subsequent one being of the same type.
- Stratifies risk as in the MESS study, taking account of EEG, MRI, neurological deficit and cognitive impairment.
- Uses more modern drugs – nowadays lamotrigine and levetiracetam are common first-line agents, as opposed to carbamazepine and valproate which were the drugs that mainly featured in the MESS study. While these are admittedly not clearly more efficacious, they are better tolerated.
- Includes an analysis of the side effects of drugs in those randomised to treatment, and the quality of life impact of these side effects and of the “inconvenience” factor of taking regular medication.
Given the current lack of clear data, we are left with clinical judgement and patient preference.
My practice with regard to a patient who has just had a generalised tonic clonic seizure is largely to ignore the data from MESS indicating that treating a first seizure non-significantly increases risk when the EEG and neurological examination are normal. How much were the data distorted by being randomised 3 months after the seizure? How many in this category had a complex partial seizure? A particular problem is that often I am not going to get an EEG within a week of the seizure, so a major risk stratification factor is unknown at the most important time to start treatment. I quote the FIRST trial as a “worst case scenario”, something like:
The risks of recurrence could be as high as 41% over the next year and medication could reduce this to 17%. However, given your neurological examination and imaging (and possibly EEG) are normal, and there is no particular evidence of a recurrent epileptic syndrome (e.g. clear family history, developmental delay, juvenile myoclonic epilepsy), the risk may be appreciably lower and the benefit of medication therefore appreciably less. The risk, which includes a slight risk of sudden death as a result of a second seizure, must be balanced against the risk of side effects of taking medication.
Particular factors relevant for you might be the further 12-month driving ban after a subsequent seizure, and teratogenic risk of drugs if you fall pregnant while taking them. (Though lamotrigine and levetiracetam have rather favourable teratogenic risk profiles.)
Then, when it comes to stopping medication, as this should really be addressed before starting:
Since you have only had one seizure, we would empirically consider you in the generally accepted “best category in whom one would initially treat” and advise at least 2 years treatment assuming no further seizures. This 2 year figure is somewhat arbitrary, reflecting that FIRST demonstrated continued risk reduction two years after starting medication but did not investigate a longer period.
If the patient has had a single complex partial seizure and no risk factors, I would explain:
For this relatively minor seizure type there is a lack of evidence for treatment and most patients are not treated. Only if you are very keen on treatment, e.g. regarding driving, would I offer it to you after counselling on potential drug side effects.
If the patient is in the medium or high risk category according to the stratification of the MESS data, in other words neurological deficit, developmental delay, cognitive impairment, features of an epileptic syndrome, or if I have an EEG already and it is abnormal, or perhaps an epileptogenic lesion on an MRI scan to boot, I will tend to use the MESS data:
A potentially risky time for seizure recurrence is in the next 3 months. Even if you do get to three months without a seizure a major study has shown that the risk of a second seizure by one year is 35% and medication may reduce this to 24% (or for the high risk category 59% to 36%). Given these risks, and the slight possibility of death from a seizure, I would advise treatment despite the potential risks of drug side effects unless you had any particular issues.
And for stopping medication again:
The long-term 5+ year follow-up in the MESS study indicated that many patients go into seizure remission at this time after their first seizure, whether or not they started on medication initially or had seizures during this time, but those who were initially treated were less likely to have seizures in getting to that 5-year milestone. Furthermore, another study (though on patients who had had more than one seizure) showed that antiepileptic drugs may still reduce the risk of a recurrence over the subsequent 12 months if you have gone up to, but not beyond, 5 years without a seizure. Even if you remain seizure-free, I therefore generally recommend 5 years of treatment before slow medication withdrawal.
If the first presentation was with status, the risk of recurrence is not much greater but the risk of recurrent status is greater and so I would advise at least a 5 year seizure-free period before withdrawal even if no risk factors. And, moving away from the single seizure scenario, if the patient has had many seizures before the seizure-free interval, or evidence of an ongoing epileptic syndrome, even beyond 5 years seizure-free I counsel that there is always a risk of recurrence and being on epileptics may reduce this risk though they have not been proven to do so.
If one happens to see the patient for the first time at around 3 months after the event, and one has an EEG, then I think one might directly apply the MESS reanalysis of stratification of risk, namely to recommend treatment only if the patient is not in the lowest risk category. However, if the seizure was generalised tonic clonic, I am still uncertain about the applicability of that study, and I counsel the patient that while there is no clear evidence for treatment from clinical trials there are still arguments for as well as against treatment.
Of course, all these recommendations would only be a basis for discussion. Some patients may be focussed on taking a medication for any possible benefit to minimise risk of extended driving bans or sudden unexpected death in epilepsy. Others may not want to risk drug side effects unless they are of proven benefit or there is any possible risk of teratogenicity (despite the risk that a generalised tonic-clonic seizure in a mother poses to her unborn baby). I do counsel strongly that if one does embark on medication treatment for an unprovoked seizure there is little point in taking it for a period of less than 2 years. I also counsel them up front about the UK’s 3-month recommended period off driving at a future time of withdrawal of medication. Even this relatively short time off driving during this potentially risky period immediately after drug withdrawal could have important connotations for some patients who have been back driving again for 18 months.
Pingback: Journal Club Review: Driving after a Single Seizure | Neurology Online Journal Club